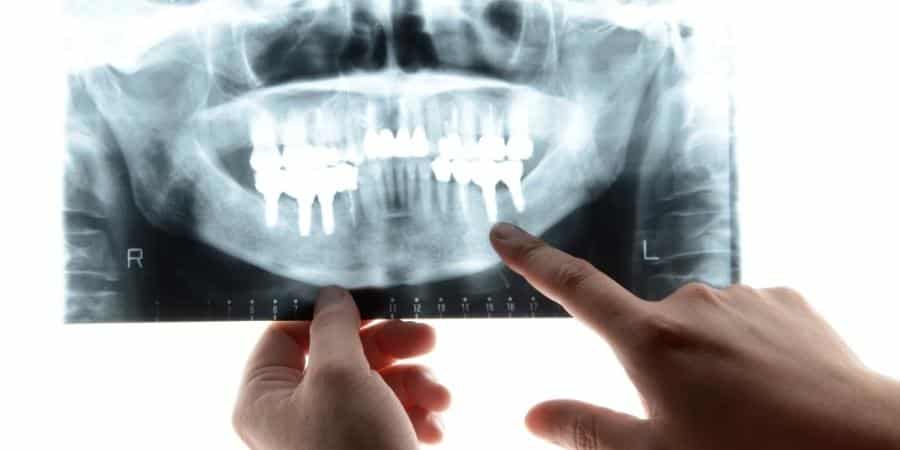
Ameloblastoma is a disorder of the jaw involving abnormal tissue growth. It is a very rare (about 1% of all jaw tumors) and slow-growing tumor that develops in the space behind our molar or back teeth. Ameloblastoma can occur over a broad age range, most commonly affecting patients between the ages of 20 and 40. Unfortunately, the causes of this disorder are not well understood to date. Studies estimate that every year 1 in 1 million people is diagnosed with ameloblastoma, and while it can become cancerous, this is quite rare and occurs in only 1% of cases.
As tissues affected by ameloblastoma are those responsible for tooth development, when left untreated it can result in damage to the jawbone and other parts of the mouth, causing severe configuration.
While surgery is typically used to treat ameloblastoma, it often involves the removal of healthy tissues around the tumor to lower the chances of recurrence, as a high recurrence rate is a serious issue. Specifically, large ameloblastomas are known to be associated with increased rates of recurrence.
Accordingly, many require reconstructive surgery to replace the bone and tissues removed and prevent visible disfigurement. As the face is one of the most complex parts of the human body, restoring its tissues presents a significant challenge, and patients are often inflicted with functional impairment and aesthetic deformities.
An Innovative Neoadjuvant Treatment for Ameloblastoma
BRAF (B-raf proto-oncogene serine/threonine kinase) is a protein responsible for cell proliferation. BRAF gene mutations have been identified in ameloblastomas, with one mutation (BRAF V600E) occurring frequently. This finding strongly suggests the possibility of targeted therapy for patients with ameloblastoma.
According to a recent Sheba study, selective inhibitors of the mutated BRAF gene given prior to a surgical procedure have not only reduced the size of tumors but were also found to aid jaw bone self-recovery, allowing it to almost regain its pre-disorder state.
The new treatment was indicated to be effective against advanced-stage ameloblastoma in children and young adults, and as such, presents the first successful upfront rational therapy for ameloblastoma in young patients.
In light of the significance of the human face as both a biological organ and what embodies our personal identity, vital to both our physical and social lives, anti-BRAF inhibitor therapy could prove to have a profound impact on the quality of life of patients, sparing them the trauma of aesthetic deformities as well as functional and psychological impairments.
The innovative neoadjuvant treatment has recently been granted approval by the FDA, and is available in only a few medical institutions worldwide, including Sheba Medical Center.